Biomolecular Motors on Nanostructures (WP6)
The Delft group has extensive experience in nanofabrication and is now moving into single-molecule
studies of motor enzymes. For a start, we will make linear extended nanostructures that will act as an
optical grid. This connects to recent work from the Paris group who has developed a new technique to
characterize single kinesin motors without any external force and at unprecented bandwidth allowing in
principle microsecond time resolution.
Currently this involves the use of optical-interference patterns,
but this will be replaced by a solid-state nanofabricated structure, which will allow a wide range of
spacings in the pattern down to the few 10 nm range, and an improved stability. Kinesin mobility will then
be measured at the single molecule limit. The technique combines nm spatial and ?s time resolution.
New physics relating to the mechanism of kinesin motion is expected.
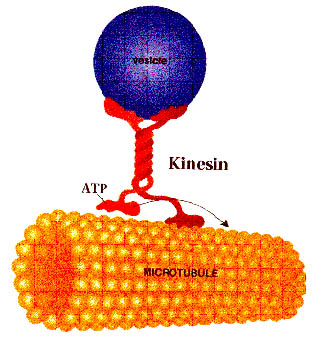
The kinesin enzym walking on a microtubule in presence of ATP
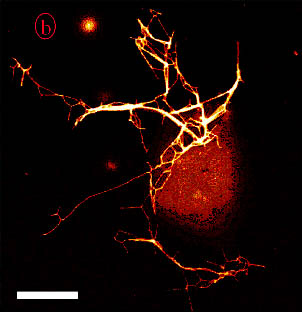
A giant liposome coated with kinesins. In presence of microtubules and ATP, kinesins pull out a network of lipid nanotubes


